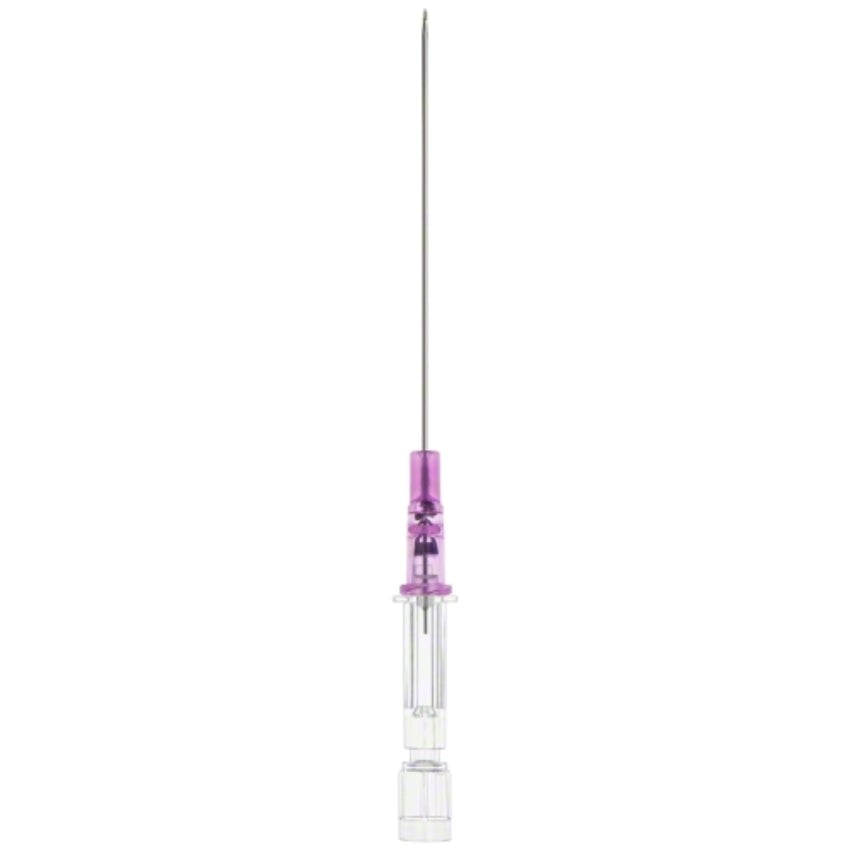
The Introcan Safety® Deep Access IV Catheters are designed to facilitate ultrasound-guided procedures where access to deeper veins is desired. These types of IV procedures are usually required for patients with difficult vascular access, including overweight/obese patients. Short PIVCs have been associated with several complications related to deeper vessels, such as catheter-related thrombophlebitis, dislodgment, and infiltration. The Introcan Safety Deep Access IV Catheters are designed to allow clinicians to leave more catheter in the vessel and help reduce the risk of complications.
Features
- Visible under ultrasound. The needle tip and catheter are visible under ultrasound, providing a visualization aid during the insertion process.
- Made using polyurethane material. Polyurethane and longer lengths associated with extending the duration of therapy.
- Designed to prevent accidental needle sticks. The fully automatic passive safety shield requires no manual activation, CANNOT BE BYPASSED, and prevents needle reinsertions.
Sold by
Introcan Safety® Deep Access IV Catheters, 20GA, 2.5in. [200/case]